Dr. Marissa Carter has posted a new blog about the latest COVID-19 news, including a vaccine update.
Vaccines
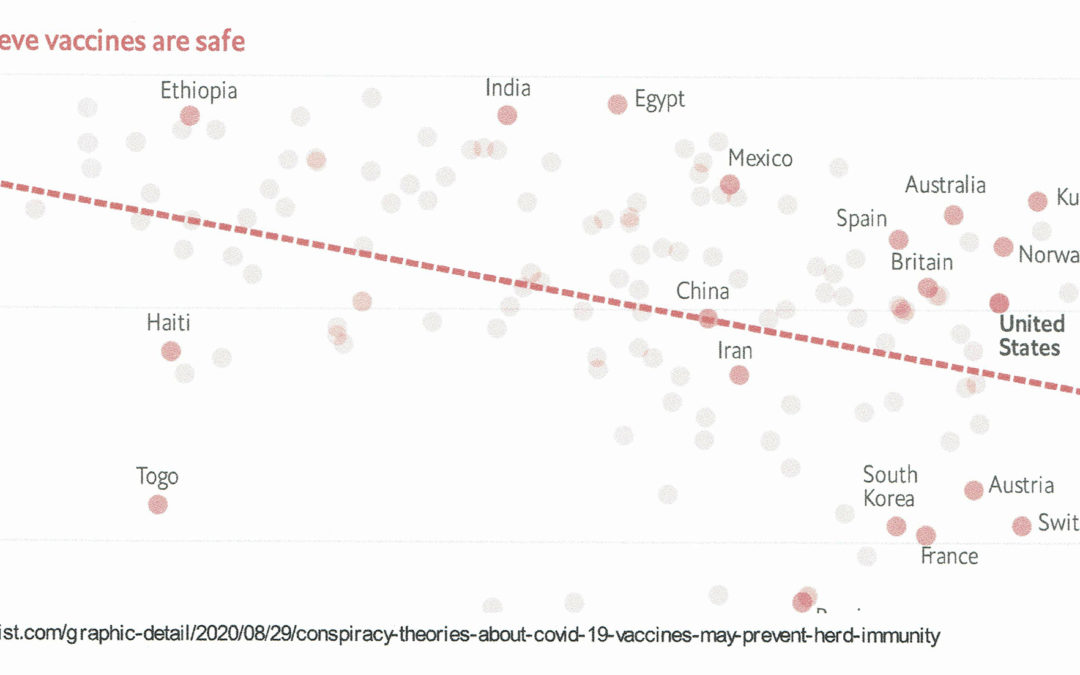
It’s been an interesting week! First, there have been a number of reports that both the Russians and Chinese have started vaccination programs in their own populations without waiting for phase 3 results of the vaccines. The Russian effort, called Sputnik 5, is based on a recombinant adenovirus type 26 (rAd26) vector and a recombinant adenovirus type 5 (rAd5) vector, both carrying the gene for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike glycoprotein (rAd26-S and rAd5-S). Injections are given 3 weeks apart. The Lancet just published the phase 1/2 results of the vaccine trial. The journal also provided an accompanying editorial and although the number of participants in the trial was small (N=76) and they were primarily young people, the results were generally good. Importantly, the vaccine is available in two versions: frozen and lyophilized, which must be stored at -18⁰C and from 2 to 8⁰C, respectively. That said, this trial is only the first step; much more research will need to be conducted to understand its safety and efficacy, but evidently president Putin has some faith in it as it was reportedly given to his own daughter.

Dr. Fife is a world renowned wound care physician dedicated to improving patient outcomes through quality driven care. Please visit my blog at CarolineFifeMD.com and my Youtube channel at https://www.youtube.com/c/carolinefifemd/videos
The opinions, comments, and content expressed or implied in my statements are solely my own and do not necessarily reflect the position or views of Intellicure or any of the boards on which I serve.



It will be a long time before I trust ANYTHING published in the Lancet