For many years I was the director of a very large lymphedema clinic at the Texas Medical Center. I worked with 6 fabulous manual lymphatic drainage (MLD) therapists. The therapists and the patients taught me nearly everything I know about lymphedema because for many years, no one had ever actually seen the lymphatics working in real time. Our understanding was entirely limited to what we could see with our naked eyes, and what they could feel. Then I started working with Dr. Eva Sevick who developed real-time, near-infrared fluorescence lymphatic imaging. As much as I trusted my fantastic therapists, occasionally they would tell me things based on palpation, and I would say skeptically, “How can you ‘feel’ that?” Well, real-time imaging has shown that they were right about nearly everything they said. As only one example, the therapists used to tell me that they could feel preclinical lymphedema in patients after breast cancer, long before the patients developed clinically obvious lymphedema. They were right. Check out this paper, just out in Breast Cancer Research and Treatment entitled, “Prediction of breast cancer-related lymphedema by dermal backflow detected with near-infrared fluorescence lymphatic imaging.”
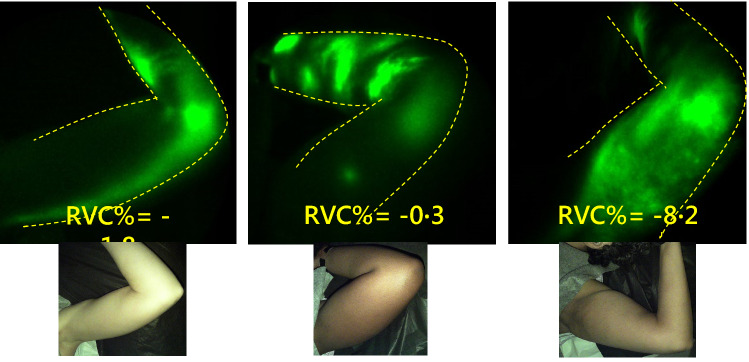
NIRF-LI images of the left axilla at 6 months after RT in study subject number 3 (left), number 12 (middle), and number 16 (right). Dermal backflow is apparent, despite the corresponding arm swelling/relative volume change (RVC%) values being − 1.8, − 0.3, and − 8.2 (left to right)
Mild breast cancer-related lymphedema (BCRL) is clinically defined as a 5%–10% increase in arm volume, typically measured no earlier than 3–6 months after locoregional treatment. If diagnosed and treated early, the outcomes of lymphedema are better. However, treatment is typically delayed until arm swelling is obvious. We investigated whether near-infrared fluorescence lymphatic imaging (NIRF-LI) surveillance could identify the early onset of peripheral lymphatic dysfunction as a predictor of BCRL.
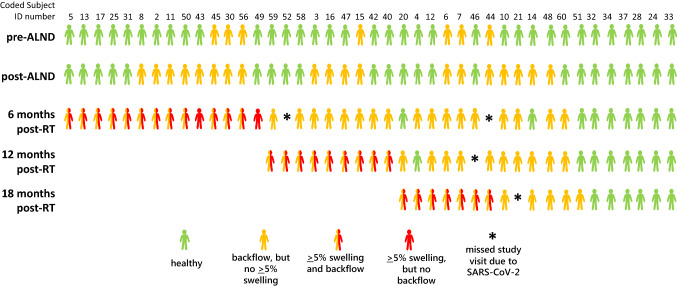
Modified swimmer plot denoting, at each surveillance visit, the absence of lymphatic dysfunction (green figures), appearance of dermal backflow (yellow), and objective diagnoses of breast cancer-related lymphedema (BCRL) from ≥ 5% RVC on the affected-side arm at 6 months after radiotherapy (RT) or later (red figures). Half-yellow/half-red figures represent subjects who displayed after-RT backflow and ≥ 5% swelling at the same visit. Asterisks denote clinical study interruption due to the SARS-CoV-2 pandemic. Blank spots after subjects developed BCRL/ ≥ 5% arm swelling do not represent subjects dropping out of the study—blank spots at 12 and 18 months post-RT allow quick visualization of BCRL onset at or after 6 months post-RT
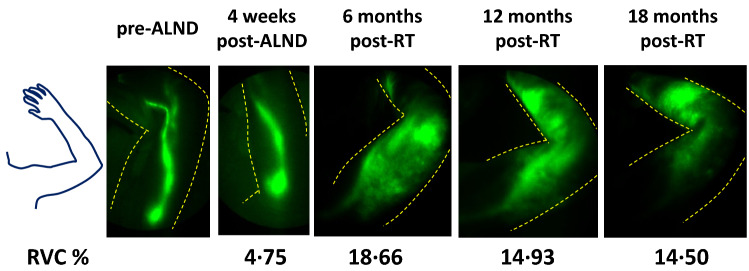
Subjects with locally advanced breast cancer who received axillary lymph node dissection and regional nodal radiotherapy (RT) were followed serially, before surgery, 4–8 weeks after surgery, and 6, 12, and 18 months after radiotherapy. Arm volume was measured by perometry, and lymphatic (dys) function was assessed by NIRF-LI. By 18 months after RT, 30 of 42 study subjects (71%) developed mild–moderate BCRL (i.e., ≥ 5% arm swelling relative to baseline), all manifested by visible “dermal backflow” of lymph into lymphatic capillaries or interstitial spaces with IC green injection. Dermal backflow had an 83% positive predictive value and 86% negative predictive value for BCRL, with a sensitivity of 97%, specificity of 50%, accuracy of 83%, positive likelihood ratio of 1.93, negative likelihood ratio of 0.07, and odds ratio of 29.00. Dermal backflow appeared on average 8.3 months, but up to 23 months, before the onset of mild BCRL. The study indicates that breast cancer related lymphedema can be predicted by dermal backflow under IC green imagine, which often appears months before arm swelling, enabling early treatment before the onset of edema and irreversible tissue changes.
My therapists said that they could “feel” these early changes before they were visible. Now the rest of us can see them thanks to near-infrared fluorescence lymphatic imaging.

Dr. Fife is a world renowned wound care physician dedicated to improving patient outcomes through quality driven care. Please visit my blog at CarolineFifeMD.com and my Youtube channel at https://www.youtube.com/c/carolinefifemd/videos
The opinions, comments, and content expressed or implied in my statements are solely my own and do not necessarily reflect the position or views of Intellicure or any of the boards on which I serve.



Great work!! – has this data been published elsewhere? If so, where?
Also, any idea when NIFLI will be more readily available?
Wow, that’s amazing! Now to get it into real time use!!