I did my first prospective clinical trial in 1997 and despite having a waiting room packed with patients, had to put ads in the Houston paper and go on radio talk shows to find patients HEALTHY enough with wounds superficial enough to participate in the trial. (Yes, in 1997, before the Internet, people read the newspaper and listened to the radio…)
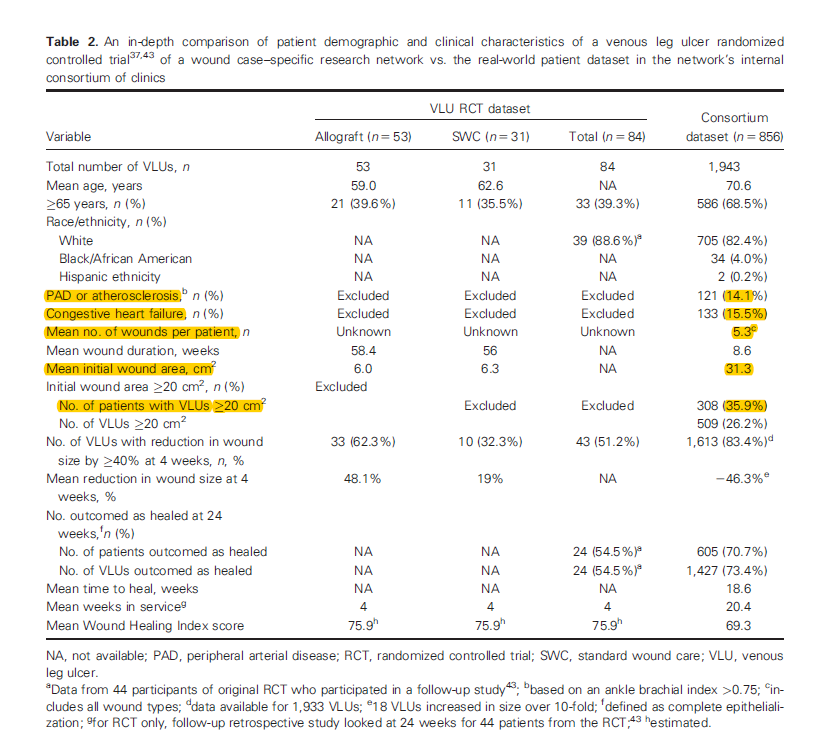
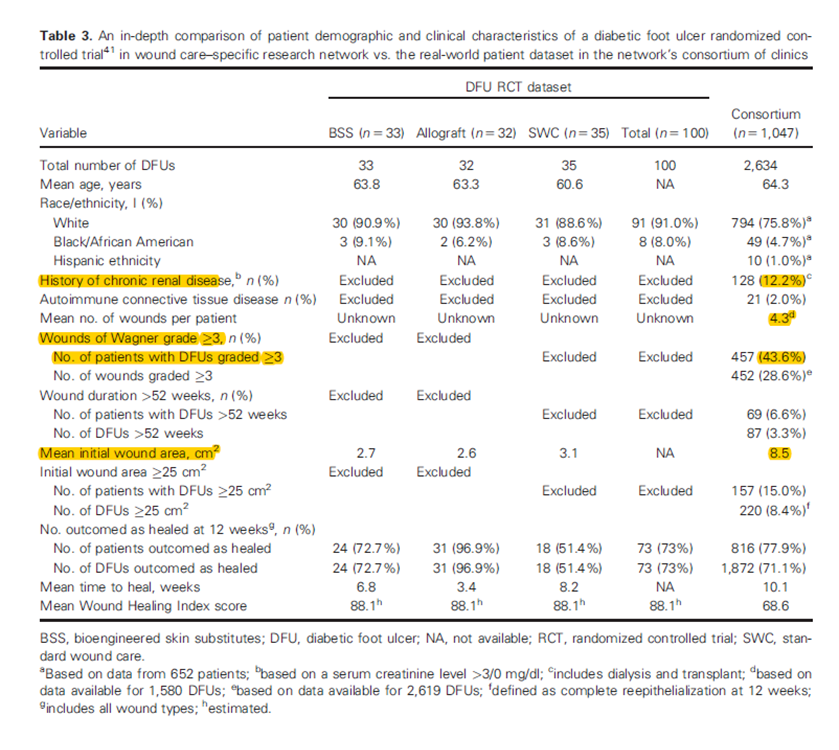
In 2017, we looked at this in a consortium of clinics being managed by the Serena Group that were also performing several prospective trials in diabetic foot ulcers (DFUs) and venous leg ulcers (VLUs). We compared the characteristics of real patients/wounds being treated in those clinics with the exclusion criteria for subjects enrolled in the trials they were performing (see tables).
The real world DFUs were much worse and the real patients were far sicker. The real VLUs were larger and those patients were sicker than the subjects enrolled. Dr. Marissa Carter and I began talking about this disconnect in 2009 when we performed a study demonstrating that the majority of patients seen in wound centers would have been excluded from participation in every prospective clinical trial that had been published over the previous decade.
Check out the podcast about this ongoing problem – an interview with Deb Borfitz. I am grateful for Deb’s interest in this issue.
A news article about the realities of chronic wound research was just released in Clinical Research News.
The non-generalizability of clinical trials is the reason Intellicure has spent the last 25 years developing real world evidence to the point it can be reliably used to understand what really happens to patients with chronic wounds and product effectiveness in the real world.

Dr. Fife is a world renowned wound care physician dedicated to improving patient outcomes through quality driven care. Please visit my blog at CarolineFifeMD.com and my Youtube channel at https://www.youtube.com/c/carolinefifemd/videos
The opinions, comments, and content expressed or implied in my statements are solely my own and do not necessarily reflect the position or views of Intellicure or any of the boards on which I serve.