In February of 2019, CMS approved hyperbaric oxygen therapy (HBOT) as a topic for audit by the RACs (Recovery Audit Contractors) (see links below). I have not heard of any hospital getting a RAC audit, perhaps because of the COVID crisis, but it doesn’t appear that the challenges of COVID-19 will prevent them from going forward.
I am a devoted follower of attorney Knicole Emanuel’s blog. Her practice concentrates on Medicare and Medicaid regulatory compliance litigation. Her most recent post is an alert that Medicare is resuming audits despite the declared public health emergency (“PHE”) for the COVID pandemic. The PHE regulations are scheduled to expire July 24, 2020, although there is strong evidence that they will be extended to Dec. 1, 2020. Nevertheless, RAC and MAC audits which might resume in August 2020 (meaning, next month).
As an aside, Ms. Emanuel describes her experience with “COVID-virtual trials,” which has been largely positive. Another issue addressed in the CMS’ July 2020 FAQs concerned the inability to collect signatures for certain Durable Medical Equipment (DME) supplies – which is highly relevant to wound care practitioners since many patients obtain their wound dressings through DME providers.
According to the July 2020 CMS FAQs:
“ . . .given the nature of the pandemic and the inability to collect signatures during this time, CMS will not be enforcing the signature requirement. Typically, Part B drugs and certain Durable Medical Equipment (DME) covered by Medicare require proof of delivery and/or a beneficiary’s signature. . .This exception may or may not extend until Dec. 31, 2020.”
It is important for wound care practitioners to note that the signatures referred to as not being required are those that confirm proof of DELIVERY of supplies required from the beneficiary or their designee. There is no COVID-19 regulatory relief for the signature required from the DOCTOR to order the DME supplies in the first place.
There is no Government official with the power to change paragraph indentations
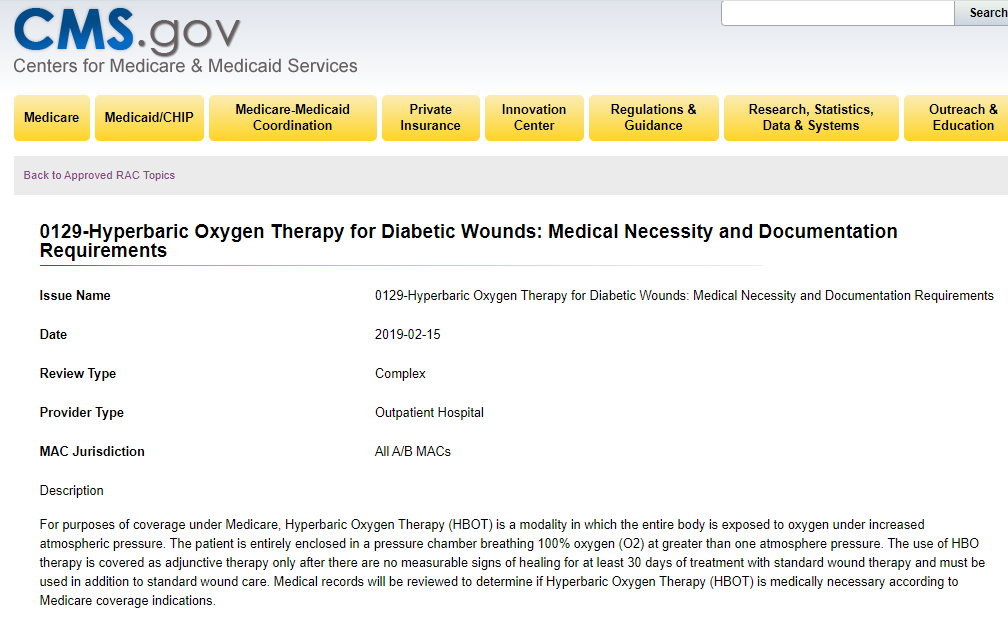
I am pointing out the possible resumption of RAC audits because in February 2019, CMS confirmed that Hyperbaric Oxygen Therapy (HBOT) was a topic for RAC audit in all Medicare regions. Here’s the link to the posting on the CMS website.
It worries me that the description (below) specifically states that they will be checking to determine whether there are no measurable signs of healing for the 30 days prior to starting HBOT. I worry about this because, while that requirement is correct for Wagner Grade 3 diabetic foot ulcers (DFUs), it is NOT a requirement for any other HBOT indication. We know of MAC audits that have recuperated HBOT reimbursement for late effects of radiation because the patient’s chart did not meet the requirements for DFUs. Helen Gelly and I have notified every Medicare official we can think of as well as various Congressmen that the DFU requirements are being improperly implemented for other HBOT indications. The reason is apparently the absence of a “paragraph indentation” after the DFU guidance and before the next HBOT indication. However, this error is now carved in stone, and we can find no official with the power to insert a paragraph indentation.
So, heads, up, if you get a RAC audit, they may be using incorrect guidance and all you can do it hope to prove that during your “virtual trial,” because we will see a cure for COVID-19 before we find an official with the authority to change a paragraph indent.
0129-Hyperbaric Oxygen Therapy for Diabetic Wounds: Medical Necessity and Documentation Requirements
Date
2019-02-15
Review Type
Complex
Provider Type
Outpatient Hospital
MAC Jurisdiction
All A/B MACs
…The use of HBO therapy is covered as adjunctive therapy only after there are no measurable signs of healing for at least 30 days of treatment with standard wound therapy and must be used in addition to standard wound care. Medical records will be reviewed to determine if Hyperbaric Oxygen Therapy (HBOT) is medically necessary according to Medicare coverage indications.
Affected Codes
G0277

Dr. Fife is a world renowned wound care physician dedicated to improving patient outcomes through quality driven care. Please visit my blog at CarolineFifeMD.com and my Youtube channel at https://www.youtube.com/c/carolinefifemd/videos
The opinions, comments, and content expressed or implied in my statements are solely my own and do not necessarily reflect the position or views of Intellicure or any of the boards on which I serve.



My team was presented with the same issue of the DFU criteria being applied to other HBO indications in round 1 of our HBO TPE audit with Noridian. We were able to find the following letter from the OIG and share it with our Noridian assigned auditor. It helped clear up the misinterpretation of the NCD guideline and I hope this will help others.
https://oig.hhs.gov/about-oig/about-us/files/letter-aawcm.pdf